 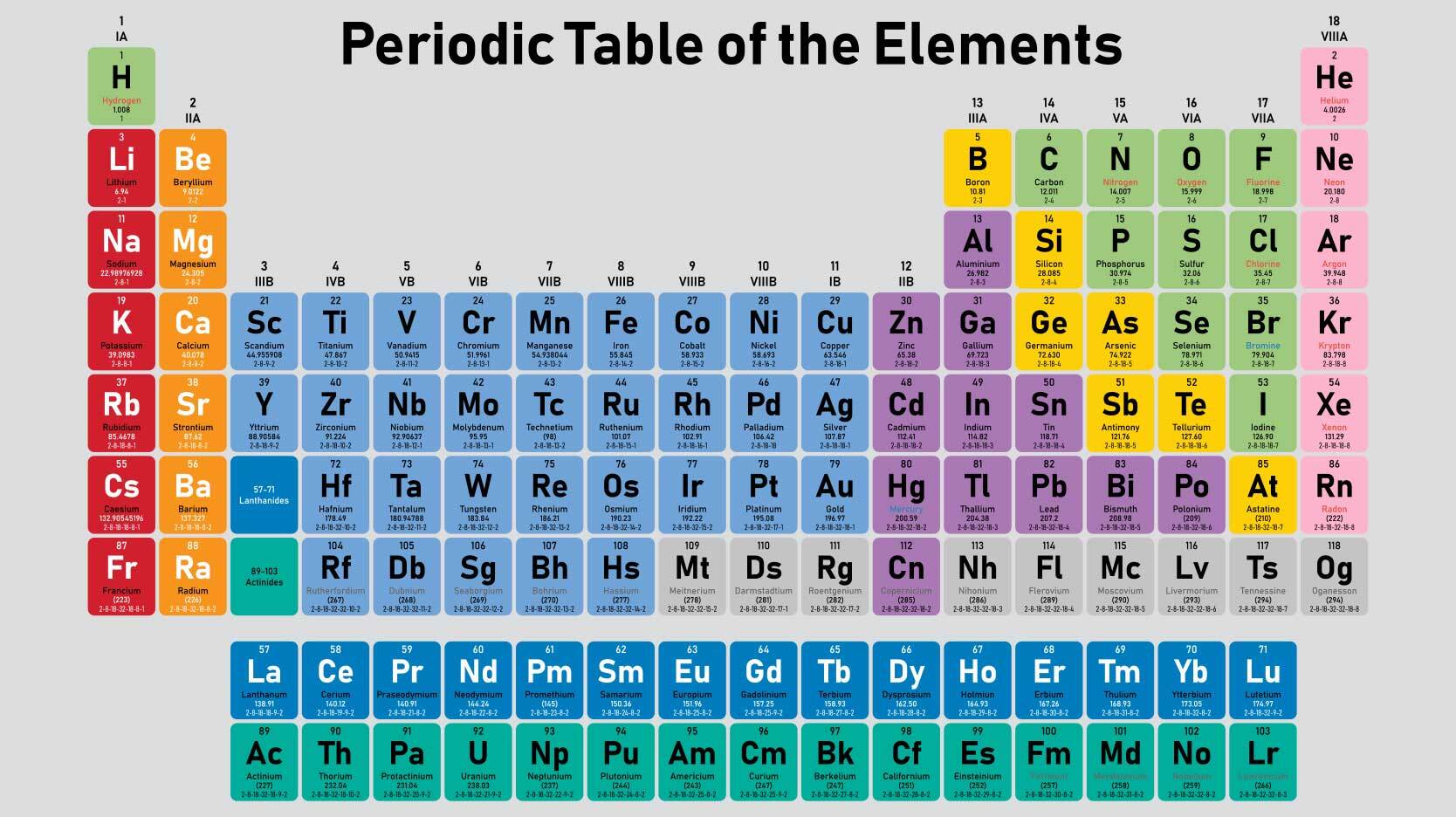
The 'noble gas' or 'inert gas' elements only become ions under very extreme conditions: most times they have no charge. If, on the other hand, an atom loses electrons, it will end up as a positive ion, with a charge of #+1#, #+2# and so on.Īll elements can become ions, but those on the left side of the periodic table tend to become positive ions while those on the right side become negative ions. One additional electron leads to a #-1# charge, two to #-2# and so on. 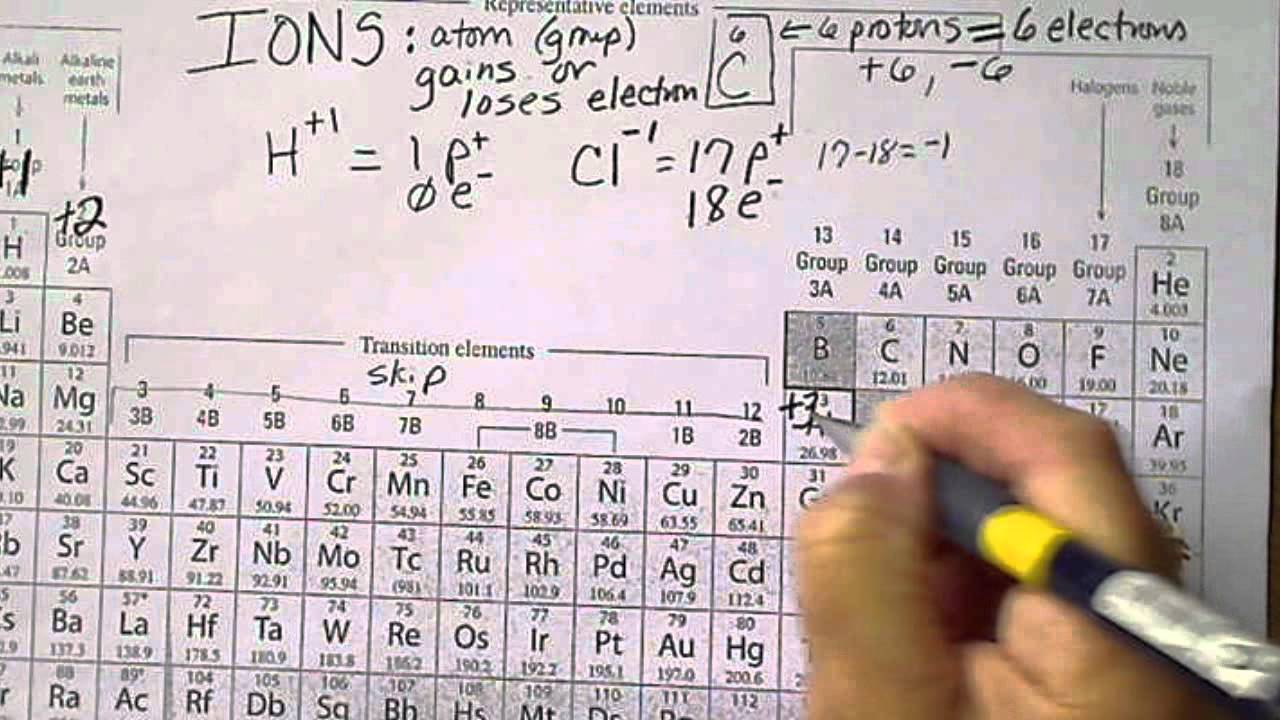
If an element gains additional electrons, it will become a negative ion. If the atom has the same number of electrons orbiting the nucleus as protons in the nucleus it will have an overall electric charge of #0#, since protons have a charge of #+1# units and electrons have a charge of #-1# units. This number is the ' atomic number', and it is the number of protons in the nucleus that makes something a particular element: 1 for hydrogen, 8 for oxygen, 17 for chlorine and so on. So that would be a nitrogen ion that you would denote like that.Each element has a particular number of protons in its nucleus. Ion Charge from Periodic Table Alkali metals all have one valence electron, which they lose to form ions with +1 charge. Periodic table of elements, we can see that if you have seven protons, by definition you are And if you wanted to writeĭown what ion that is, once it's gonna go back to the And so I would say you'd often denote that as saying a three minus charge. So seven minus 10, that wouldīe equal to negative three. That's why you're subtracting, you subtract out the electrons. Identify the location of the elements in the periodic table. For example, a sodium atom usually has a +1 charge, while oxygen most often has a -2 charge, and chlorine usually has a -1 charge. The valence of an atom is its oxidation state. Of protons, seven, which are the positive charges, and you subtract out the negative charges. This printable periodic table contains the atomic number, element symbol, element name, atomic weights and most common valence charges. And if you wanna figure out the charge, you just take the number That indeed would be an ion because it has a different number of protons than it does electrons. Ions formed from elements in Group IA of the periodic table have a 1+ charge. What is the charge of an ion that has seven protons, eight neutrons, and 10 electrons, pause this video and think about what that would be. Atoms are electrically neutral because the number of protons, which carry a 1+ charge, in the nucleus of an atom is equal to the number of electrons, which carry a 1- charge, in the atom. Image credit: Wikipedia Commons, public domain. Lost two of those electrons and so then it got a positive Sodium chloride is an ionic compound made up of sodium ions and chloride ions in a crystal lattice. So then it would not be an ion, it would just be a neutral atom. Originally had 20 electrons and 20 protons. To show that it is a calcium ion, it's likely a situation maybe where the calcium With positive two charge, 20 minus 18 is positive two, and we will denote that with a two plus. So if you wanna know the netĬharge, you take the number of protons, the positive charge and subtract out the number of electrons. The anion is the sulfide ion and its charge from the location in the periodic table is 2. Number electrons, I'llĪbbreviate it right over there or I'll shorten it. Table 5.1(1) Common Cations of Metals That Form More Than One Ion. Protons which provide positive charge is 20. They are sufficiently stable that they don’t form either anions or cations easily. The element is actually how many protons it has, and that's what we have right over here. To put it another way, elements on the left side of the periodic table tend to form cations, while those on the right side form anions. So why don't you pause this video and see if you canįigure out the charge of that calcium ion. Might be useful to see where calcium sits on that And I will give you a little bit of a tip. Asked, what is the charge of a calcium ion with 18 electrons? So pause this video and see if you can work that on your own.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
